Epigraph Vol. 21 Issue 4, Fall 2019
Epilepsy medications and pregnancy: What are the issues?
Anti-epileptic drug (AED) choice in girls and women requires a long lens. A clinician must look into the future — sometimes decades — and consider the impact of the AED on any future pregnancies.
Several AEDs — most notably, valproate — pose risks to the developing fetus. Two recent studies have confirmed that of commonly used AEDs, valproate is associated with the highest risk for certain birth defects, but that valproate prescriptions are declining — as are rates of such birth defects — as more and more health care professionals are made aware of the risks.
Not all countries have guidelines for AED choice, AED monitoring, and folic acid supplementation in women with epilepsy — and the guidelines that do exist are quite variable.
Read a related article: Six ways to maximize reproductive health in women with epilepsy
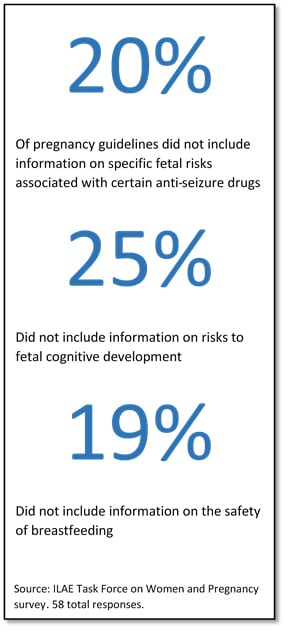
The results of a recent ILAE survey showed that 69% of countries have guidelines or recommendations for managing epilepsy during pregnancy. However, some of the guidelines had not been updated in more than a decade, and only 46 of the countries confirmed that the guidelines made specific recommendations about AED selection. In addition, the survey received information from only 77 countries, with little representation from countries in Africa and parts of Asia/Oceania.
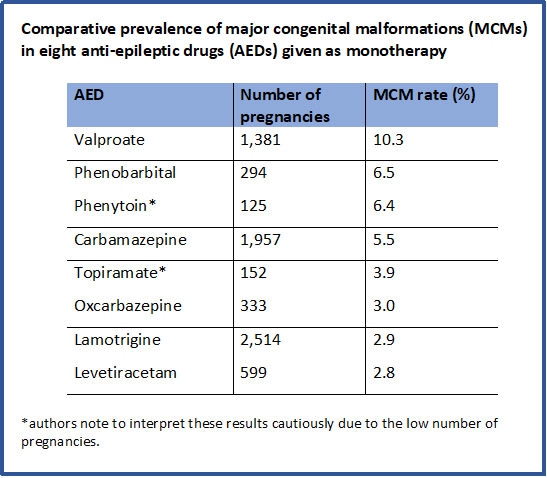
Though more than 25 AEDs are prescribed for people with epilepsy, only about one-third have any data on fetal risk from human studies. A 2018 analysis of EURAP data — including 7,355 pregnancies from 42 countries — calculated rates of major congenital malformation (MCM) associated with eight AEDs used as monotherapy. Rates were highest in women taking valproate, and lowest in those taking lamotrigine and levetiracetam (see box). Risks associated with lamotrigine, levetiracetam and oxcarbazepine were similar to risks seen in offspring not exposed to AEDs.
Multivariable analysis showed that compared with low-dose lamotrigine, the prevalence of MCMs was significantly higher for all doses of valproate and carbamazepine, as well as for phenobarbital at doses of more than 80 mg/day. Compared with levetiracetam (250-4,000 mg/day), low-dose valproate and high-dose carbamazepine were associated with an increased risk of MCMs.
Drug choice – birth defects and cognitive issues
Although the risk of birth defects from valproate is relatively well known – several regulatory bodies have restricted its use in women – the drug is sometimes the only option for keeping women seizure free, noted Torbjӧrn Tomson, professor of neurology and epileptology at Karolinska Institutet and head of the epilepsy section at Karolinska University Hospital, Stockholm. “For idiopathic generalized epilepsy, valproate is superior,” he said. “There are women that need it. But they need to be fully informed and understand what the risks are.”

Earlier this year, Tomson and colleagues published an analysis of EURAP data showing a 27% decline in MCMs in children born to women with epilepsy between 2000 and 2013. The decline mirrored a decrease in valproate prescriptions and a corresponding increase in lamotrigine and levetiracetam prescriptions.
“When we adjusted for changes in treatment, the trend disappeared, so we knew the declining malformation rate was linked to the changes in prescriptions,” he said.
The analysis showed no decrease in seizure freedom, at least on a group level. “However, we know there are individual women who cannot switch from valproate and maintain seizure control,” said Tomson. “But the availability of some newer antiseizure medications has made a difference. With 600,000 pregnancies a year in women with epilepsy, reducing the risk of malformations by 27% has a real impact.”
Valproate use also carries risks of cognitive issues and autism characteristics. Data from the NEAD study group showed that at 3 years of age and 6 years of age, children with prenatal exposure to valproate had lower IQ scores, poorer memory and lower executive functioning skills than children exposed to lamotrigine, phenytoin, or carbamazepine. Dose mattered; children exposed to the highest doses of valproate fared the worst. And a 2013 Danish registry study of more than 655,000 births showed increased risk for autism in children born to women taking valproate during pregnancy.
Children born of women taking valproate during pregnancy also tend to have poorer cognitive development. Data from the NEAD study group showed that at ages 3 years, 4.5 years, and 6 years, children with prenatal exposure to valproate had lower IQ scores, poorer memory and lower executive functioning skills than children exposed to lamotrigine, phenytoin, or carbamazepine. Dose mattered; children exposed to the highest doses of valproate fared the worst. And a 2013 Danish registry study of more than 655,000 births showed increased risk for autism in children born to women taking valproate during pregnancy.
Both the US Food and Drug Administration and the European Medicines Agency (EMA) have advised against prescribing valproate to females of childbearing age unless no effective alternative exists. In September 2018, the EMA effectively banned the use of valproate for women of childbearing age who have migraine or epilepsy, with certain exceptions:

- A woman with epilepsy already taking valproate who cannot switch to another AED can continue to take valproate. These women should receive specialist care during pregnancy.
- For new valproate prescriptions, EMA mandates the prescribing physician follow a pregnancy prevention program, which entails:
- Assessing the potential for pregnancy, and involving each woman in evaluating her circumstances and supporting informed decision making
- Administering pregnancy tests before starting epilepsy treatment, as well as during treatment (as needed)
- Counseling women about the risks of treatment with valproate
- Explaining the need for effective contraception throughout treatment
- Carrying out specialist review of treatment at least once a year
- Reviewing a new risk acknowledgment form with each woman to confirm that appropriate advice has been given and understood
Tomson says that most care providers of women with epilepsy—at least those represented in EURAP data—appear well aware of the risks of valproate. “EURAP includes 40 years of data through 2013, and during that time we saw a dramatic decline in valproate use, though the strict indications from the European Medicines Agency and the FDA came after 2013,” he said. “But does everyone who needs to know about valproate know? Of course they don’t.”
To increase awareness, ILAE’s Task Force on Women and Pregnancy is writing a series of reviews about managing pregnancy in women with epilepsy. The first review was published November 24 in Epilepsia.
AED doses, before and during pregnancy
Higher doses of several AEDs carry higher risks of birth defects; for this reason, experts note that women should be on the lowest dose possible for seizure control. Stopping AEDs altogether or switching AEDs during early pregnancy carries a risk for seizures; abrupt discontinuation of any AED is not recommended, given the risks of seizures, status epilepticus and sudden death due to epilepsy (SUDEP).

Yet many women with epilepsy still believe they should stop taking their AEDs if they become pregnant. A 2014 study found that 40% of women with epilepsy had poor medication adherence during pregnancy, and that pregnancy-related beliefs about medication were a significant factor. Clear communication with female patients is key to improving adherence and reducing the risk for seizures during pregnancy.
Due to pregnancy-related changes in drug clearance, it may be necessary to adjust AED doses during gestation. Certain drugs — such as lamotrigine and levetiracetam — appear to fluctuate more during pregnancy than other drugs, such as carbamazepine. Monthly monitoring to ensure clinically appropriate AED levels is ideal, said Page Pennell, director of research at Brigham & Women’s Hospital and professor of neurology at Harvard University. But experts agree that such monitoring is not always feasible.
“Many countries have no access to drug level monitoring,” noted Tomson, “which makes it difficult to manage women who are using those drugs most prone to pharmacokinetic changes.”
A recent survey of 642 epilepsy care providers from 81 countries found that providers other than epilepsy specialists were significantly less likely to monitor levels of lamotrigine or levetiracetam. As expected, respondents from higher-income countries were more likely to monitor blood AED levels more than once a trimester, compared with respondents from lower-income countries.
Registries and future research
Much more research is needed on the effects of AEDs during pregnancy, said Pennell. “For most AEDs we have at least some information on malformations but for the newer AEDs we have almost no information, and there’s very little long-term outcomes information,” she said. “In the future, if we can come up with other ways to predict risk — through genetic testing, biomarkers, and the like — we could personalize the approach for each person.”
The bulk of knowledge thus far often comes from pregnancy registries, noted Tomson. “It’s important to encourage people to contribute to a registry, wherever they are,” he said. “You can make a difference.”
Registry information
Australian registry
www.neuroscience.org.au/australian-epilepsy-pregnancy-register
North American registry
www.aedpregnancyregistry.org
EURAP — International registry
www.eurapinternational.org
Kerala (India) registry
email krep@sctimst.ac.in
Telephone +91 471 2524487 (weekdays 8 am – 5 pm, GMT + 5:30)
UK registry
www.epilepsyandpregnancy.co.uk
Subscribe to the ILAE Newsletter
To subscribe, please click on the button below.
Please send me information about ILAE activities and other
information of interest to the epilepsy community